Rapid rise in COVID-19 in children: Key takeaways from the AAP report on COVID-19 and kids
You may have seen the news this week that the American Academy of Pediatrics (AAP) and Children’s Hospital Association (CHA) released a new report about a rise in COVID-19 diagnoses in children. The headlines are scary — but are they accurate? We discussed the report with Boston Children’s infectious disease specialist Dr. Richard Malley to understand the data and the potential impact on families.
The report found that 339,000 children in the U.S. tested positive for COVID-19 between April 16 and July 30, 2020 — yet nearly a third of those children (97,000) were diagnosed with COVID-19 during a two-week period in late July. In fact, in the month of July alone, the number of children with the virus doubled, with more than 171,000 new reports.

“This study serves as a good reminder that kids are not immune to COVID-19,” says Dr. Malley, who was not part of the study or the report. “Even though they are at lower risk of complications compared to adults, as we see an increase in the number of kids who have COVID-19, we should expect, sadly, to see an increase in symptomatic COVID-19 and also multisystem inflammatory syndrome in children (MIS-C).” MIS-C is a delayed complication of coronavirus infection.
Children represent nearly 9 percent of all COVID-19 cases
The report summarized other key details about COVID-19 infection in children between mid-April and the end of July:
- Children represent 8.8 percent of all reported COVID-19 cases in the U.S.
- New cases of COVID-19 in children increased by 40 percent in the last two weeks of July, from 241,904 to 338,982.
- Positive test results varied widely with data from eight states, showing that between 3.6 and 17.8 percent of children tested had a positive COVID-19 test.
- Between 0.6 and 8.9 percent of children with COVID-19 in 20 states and New York City were hospitalized.
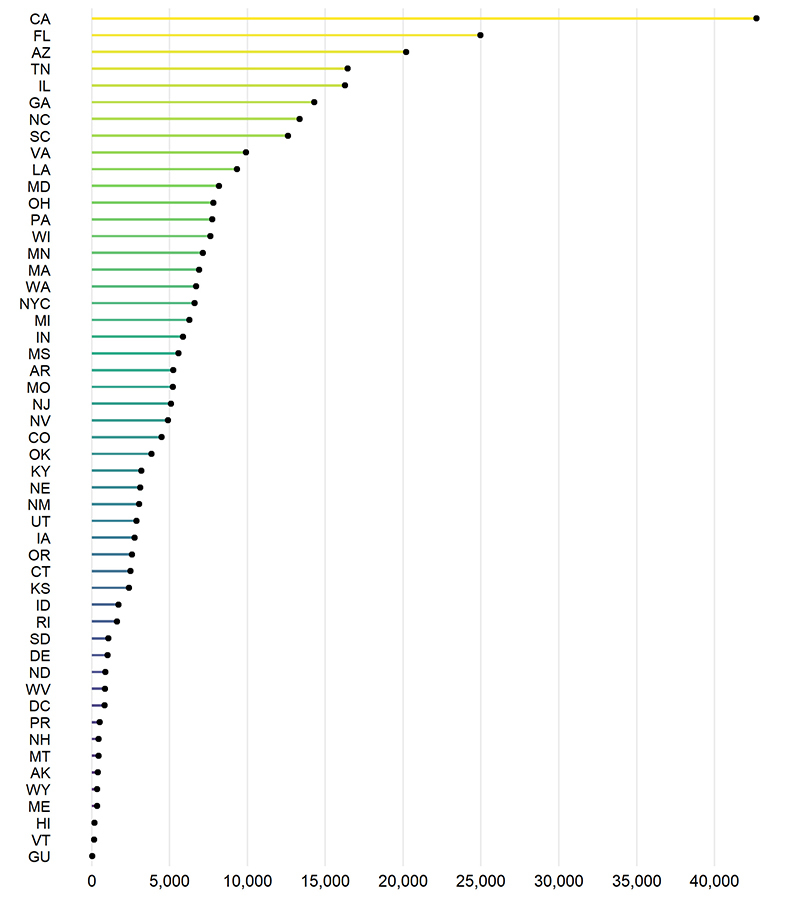
The American South and West appeared to be particularly affected: California, Florida, Arizona, Alabama, Tennessee, and Illinois all reported more than 15,000 COVID-19 cases in children. Half of the states studied (including Massachusetts) reported more than 5,000 child cases; 10 states reported fewer than 1,000 child cases.
The reasons for the increase in COVID-19 reports in children are not clear; it may represent a combination of increased testing and exposure in children. “COVID-19 is a disease of mobility, meaning that as kids’ exposure to others increases — like in overnight camps or other close-contact activities — the more they are going to get COVID-19,” says Malley.
“This study serves as a good reminder that kids are not immune to COVID-19.” Richard Malley, MD
Serious complications rare, but vigilance still needed
On a positive note/the good news: The study shows that even if a child is diagnosed with COVID-19, the vast majority will not need hospitalization. But the disease isn’t benign. Between May 21 and July 30, 86 children in the U.S. died from COVID-19.
“It was recognized early on that the risk of severe disease from COVID-19 was much lower than in adults,” says Dr. Malley. “But we need to remain careful in taking steps to prevent all infections as best we can, since infected children can still suffer from serious complications and we do not know yet to what extent there may be long-term consequences of COVID-19 infections in adults or kids. ”
Read more about MIS-C research at Boston Children’s, including a national study on the condition led by our researchers.
Related Posts :
-

Parsing the promise of inosine for neurogenic bladder
Spinal cord damage — whether from traumatic injury or conditions such as spina bifida — can have a profound impact on bladder ...
-

Unveiling the hidden impact of moyamoya disease: Brain injury without symptoms
Moyamoya disease — a rare, progressive condition that narrows the brain’s blood vessels — leads to an increased risk of stroke ...
-

Forecasting the future for childhood cancer survivors
Children are much more likely to survive cancer today than 50 years ago. Unfortunately, as adults, many of them develop cardiovascular ...
-

Genomic sequencing transforms a life: Asa’s story
Asa Cibelli feels like he’s been reborn. The straight-A middle schooler plays basketball and football, does jiu jitsu, is ...