Gene therapy gets in the ring with another disease

Seeing that his mother, Kadriye, wasn’t looking, Emir Seyrek got an impish grin on his face, the kind only a two-year-old can have. He quietly dumped his bowl of dry cereal out on his bed and, with another quick look towards his mother, proceeded to pulverize the flakes to dust with his toy truck. The rest of the room burst out laughing while his mother scolded him. Despite the scolding, though, the impish grin remained.
It was hard to believe that he arrived from Turkey six months earlier fighting a host of bacterial and viral infections. Emir was born with Wiskott-Aldrich syndrome (WAS), a genetic immunodeficiency that left him with a defective immune system. He was here because he was the first patient—of two so far—to take part in an international trial of a new gene therapy treatment for WAS at Dana-Farber/Boston Children’s Cancer and Blood Disorders Center. And that day he was having his final checkup at Boston Children’s Hospital’s Clinical and Translational Study Unit before going home.
A medical journey
Occurring only in boys, WAS starts with an error in or loss of a single gene, also called WAS, on the X chromosome. Without a working WAS gene, maturing immune cells can’t react to infection, and platelets can’t develop properly.

“Boys with WAS are prone to bleeding, eczema and certain infections,” says Sung-Yun Pai, MD, a gene therapy and immunodeficiency transplant specialist at Dana-Farber/Boston Children’s who is running the U.S. arm of the gene therapy trial, along with Dana-Farber/Boston Children’s leader David Williams, MD, and Boston Children’s Hospital immunologist Luigi Notarangelo, MD. She points out that the only current cure for WAS is a hematopoietic stem cell transplant, which replaces a child’s blood and immune system with those from a healthy donor.
But not every boy with WAS can get a transplant. “It’s not always possible to find an appropriate match,” Pai explains.
Such was the case with Emir. By the time he was diagnosed with WAS, he’d been in and out of hospitals all over Turkey since he was less than a month old. Once they knew what he had, “our doctors searched all over the world for a donor,” Kadriye says. “We finally found one, but at the last moment he backed out, forcing us to start the search all over again. Then one of our doctors asked us if we’d ever heard of gene therapy.”
Lovenah Eziuku, a Nigerian émigré who lives in Texas, tells a similar story about her adopted son Chinonso, the second patient to enter the WAS trial. During a year and a half of tests and hospitals in Nigeria and the U.S., she almost lost Chinonso more than once to rampant infections and bleeding. Finally, doctors at Texas Children’s Hospital diagnosed him with WAS.
“We then searched for a year for a donor, knowing that the clock was ticking and that we were running out of time,” Lovenah says. “When it was clear that we weren’t going to find one, our doctor invited Dr. Pai to talk to us.”
Within weeks, both boys were on planes bound for Boston.
Replacing what’s broken
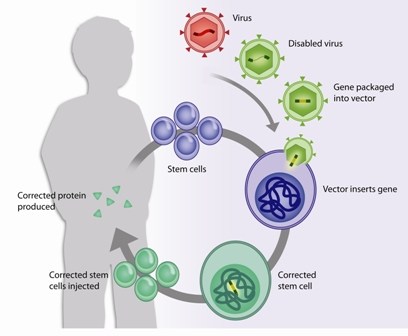
The gene therapy trial, which is also taking place in the U.K. and France, essentially attempts to replace the broken WAS gene in a patient’s bone marrow. To do so, doctors collect blood-forming (or hematopoietic) stem cells from his blood or bone marrow. In the laboratory, the cells are mixed with a vector—a virus carrying a working copy of the WAS gene. The Boston team and their collaborators are using a similar strategy in another international gene therapy trial for boys with X-linked severe combined immunodeficiency syndrome, also known as the “bubble boy” disease.
While they wait for the virus to plug the working gene into the stem cells’ DNA, the doctors give the boys chemotherapy to wipe out their existing bone marrow and blood cells. “WAS affects all cells of the immune system,” Pai explains. “We need to level the playing field and give the gene-corrected cells a chance to move into the marrow and engraft.”
Once the vector has done its job, the patient’s corrected cells are injected back into him. Then the waiting begins while the cells migrate to the marrow and, hopefully, begin to churn out new, functional immune and blood cells.

“When Emir arrived, he had had several infections and very low platelet levels,” Pai says. “After his treatment, once his white blood cells returned, his platelet count went up very quickly. And he’s carrying the corrected WAS gene now in many kinds of blood cells, which is a good sign.
“Chinonso was much sicker than Emir when he came here, with multiple active viral infections and bad eczema,” Pai continues. “Since his treatment, his cells are definitely carrying the WAS gene, and while it’s too early to tell yet whether his platelet count will get high, it’s already higher than it was before treatment. If he can get above the level that we consider the cutoff for allowing a child to play contact sports, we’ll be happy.”
Keeping a wary watch
Both boys have years of follow-up ahead of them. The trial calls for their blood to be checked for 15 years, both to make sure their newly-inserted WAS gene is working and to watch for any complications, especially leukemia. Some boys in prior WAS gene therapy trials developed leukemia later on, because the vector accidentally plugged the gene into parts of the cells’ DNA that encouraged tumor growth.
Pai and her colleagues believe the vector they are using in this trial—which differs from the one used in prior trials—should avoid those complications.
“We still have a lot of questions left to answer, especially about whether this new vector truly is safer,” Pai notes. “But we’re quite hopeful.”

But as far as the two mothers are concerned, the trial has already been a success.
“He doesn’t have eczema anymore, he’s eating better and looks better than he did before,” Kadriye said the day before they left to go back to Turkey. “He’s such a different boy than when we came here. I want to make sure he has a good childhood, grows up, and makes something of himself.”
Lovenah is excited to bring Chinonso home to her husband and three daughters in Texas, noting that last year was their third Christmas in a hospital. “Chinonso’s skin has completely cleared, he really is looking a lot better,” she says. “And he barely had any of the symptoms that can come with the chemotherapy and the treatment. He has a lot of strength, and he needs to live his life now.”
Related Posts :
-

A sickle cell first: Base editing, a new form of gene therapy, leaves Branden feeling ‘more than fine’
Though he doesn’t remember it, Branden Baptiste had his first sickle cell crisis at age 2. Through elementary school, he ...
-

A universal gene therapy for Diamond-Blackfan anemia is poised for clinical testing
Diamond-Blackfan anemia (DBA), first described at Boston Children’s Hospital in 1938, is a rare blood disorder in which the bone ...
-

Cerebral adrenoleukodystrophy: How the Cooksons dodged a devastating disease
Heather Cookson believes that if she hadn’t insisted her son Ricky get a brain MRI to investigate his frequent ...
-

Gene therapy for adrenoleukodystrophy: Studies find both risks and benefits
Cerebral adrenoleukodystrophy (CALD), portrayed in the film Lorenzo’s Oil, is a devastating disorder caused by a mutation on the ...