Getting a grip on genetic loops
A new discovery about the spatial orientation and physical interactions of our genes provides a promising step forward in our ability to design custom antibodies. This, in turn, could revolutionize the fields of vaccine development and infection control.
“We are beginning to understand the full biological impact that the physical structure and movement of our genes play in regulating health and development,” says Frederick Alt, PhD, director of the Boston Children’s Hospital Program in Cellular and Molecular Medicine (PCMM) and the senior author of the new study, published in the latest issue of Cell.
Recent years of research by Alt and others in the field of molecular biology have revealed that it’s not just our genes themselves that determine health and disease states. It’s also the three-dimensional arrangement of our genes that plays a role in keeping genetic harmony. Failure of these structures may trigger genetic mutations or genome rearrangements leading to catastrophe.
The importance of genetic loops
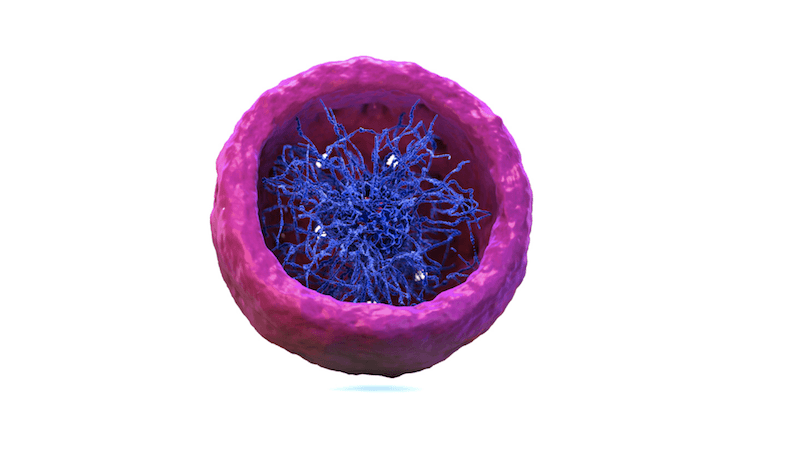
Crammed inside the nucleus, chromatin, the chains of DNA and proteins that make up our chromosomes, is arranged in extensive loop arrangements. These loop configurations physically confine segments of genes that ought to work together in a close proximity to one another, increasingly their ability to work in tandem.
“All the genes contained inside one loop have a greater than random chance of coming together,” says Suvi Jain, PhD, a postdoctoral researcher in Alt’s lab and a co-first author on the study.
Meanwhile, genes that ought to stay apart remain blocked from reaching each other, held physically apart inside our chromosomes by the loop structures of our chromatin.
But while many chromatin loops are hardwired into certain formations throughout all our cells, it turns out that some types of cells, such as certain immune cells, are more prone to re-arrangement of these loops.
Chromatin loops of the immune system
The adaptive immune system — which defends us from viruses, bacteria and other invaders — operates on a highly sophisticated system that allows genes to recombine. These gene recombinations enable our immune cells to produce antibodies against the millions of different threats that our bodies may encounter over a lifetime.
A portion of the genes that encode for antibody production is formed by rearrangement of gene segments called VDJ genes in a process that acts like a slot machine, capable of creating an infinite number of genetic combinations. Within immune cells, these tailor-made instructions enable specific antibodies to be synthesized to combat each type of pathogen that exists.
“Different V and J gene segments always come together like a sandwich on either end of a D gene segment, forming what’s called a V(D)J combination, the diversity of which underlies the production of unique antibodies,” explains Alt, who is also the Charles A. Janeway Professor of Pediatrics at Harvard Medical School.
Unlocking our understanding of V(D)J recombination control and ultimately harnessing it to design custom antibodies could herald a major breakthrough in modern vaccine strategies and development of therapeutic antibodies.
Discovering how loops form
By developing new high-throughput sequencing techniques, Alt and his team have discovered the importance that a certain small chunk of the genome — called CTCF binding elements or “CBE” for short — play in the formation of V(D)J chromatin loops and how this process guides the generation of diverse antibody genes.
The traditional school of thought was that gene recombination centers, made up of enzymes called RAGs, find distant V, D and J gene segments by a random diffusion process. However, Alt lab’s recent studies have shown that long stretches of chromatin containing these gene segments are actually pulled past the RAG complex in a conveyer-belt-like process, allowing RAG to scan the passing chromatin for its targets.
Alt’s team discovered that during scanning, CBE sequences located near V gene segments provide a bait that pulls chromatin into the RAG gene recombination center, like rope slipping through the neck of a lasso. When the RAG encounters a V gene’s CBE, scanning slows down, leading to the formation of a new loop, a process that molecular biologists call chromatin loop extrusion.
“Loop extrusion is currently a hot field in chromatin biology,” says Zhaoqing Ba, PhD, a postdoctoral researcher in Alt’s lab and a co-first author on the study.
Pioneering research into loop extrusion
To make their discovery, Alt’s team genetically modified CBEs in mice to see how the presence or absence of the “slow down” signal affected V(D)J combinations. When the nearest V segment was missing its CBE neighbor, the RAG enzyme instead zoomed along and selected the next-closest V segment with an intact CBE nearby. As they deleted CBEs farther and farther out, the process continued, with the RAG recombination center scanning along until matching up with the next-closest V gene that had a CBE nearby.
Knowledge of this process now unlocks the ability to force construction of new V(D)J combinations at the rate of millions of new variants. By generating this magnitude of variations in mice, it stands to speed up screening for HIV vaccines or identify new therapeutic antibodies.
The findings also offer major new insights into the epigenetic functions of loop extrusions. Alt and his team believe that environmental or genetic factors that trigger variations in the formation of chromatin loops could contribute to diverse diseases. Understanding the mechanisms of loop extrusion could therefore lead to the development of new classes of therapeutics.
“This is a window into epigenetics at a whole new level,” Alt says.
Related Posts :
-

New research sheds light on the genetic roots of amblyopia
For decades, amblyopia has been considered a disorder primarily caused by abnormal visual experiences early in life. But new research ...
-

Thanks to Carter and his family, people are talking about spastic paraplegia
Nine-year-old Carter may be the most devoted — and popular — sports fan in his Connecticut town. “He loves all sports,” ...
-

A new druggable cancer target: RNA-binding proteins on the cell surface
In 2021, research led by Ryan Flynn, MD, PhD, and his mentor, Nobel laureate Carolyn Bertozzi, PhD, opened a new chapter ...
-

Genetic causes of congenital diarrhea and enteropathy come into focus
Congenital diarrheas and enteropathies are rare and devastating for infants and children. Treatments have consisted mainly of fluid and nutritional ...