New strategies for restoring myelin on damaged nerve cells
Key takeaways
Myelin is an essential fatty substance coating surrounding nerve axons.
After injury, regenerated nerve axons usually fail to remyelinate, inhibiting nerve cell communication.
A combination treatment led to remyelination in about 60 percent of regenerated axons.
This research may have implications in other diseases associated with myelin loss, like multiple sclerosis.
Loss of myelin — the fatty substance that surrounds the axons of nerve cells — is one of the reasons nerve cells fail to recover after injury and in some diseases. Myelin acts like insulation, covering the long axon threads that enable high-speed communication between neurons. Without myelin, the neurons may not be able to coordinate well, leading to less than optimal function. In new research from the laboratory of Zhigang He, PhD, of the F.M. Kirby Neurobiology Center, scientists discovered a two-pronged approach to restore myelin on regenerated axons in a mouse model of optic nerve injury. Their findings may have implications for disease associated with myelin loss, like multiple sclerosis (MS).
“We believe that this work represents an important step towards functional restoration of cells in the adult central nervous system,” says He.
Axons fail to remyelinate after injury
In earlier research, He’s lab discovered several treatments that could promote the regeneration of damaged axons in the optic nerve, but do not restore nerve function. The reason? These regenerated axons are not myelinated. In this new study, published in Neuron, He explains why those axons fail to remyelinate after injury.

In the adult brain, myelination is carried out by cells called oligodendrocyte precursor cells (OPCs).
“We found that in injured optic nerves, OPCs fail to differentiate into mature myelination-competent oligodendrocytes,” says He, meaning they do not develop into cells capable of producing myelin and functioning normally.
He’s team discovered two reasons why. The first is that OPCs in injured nerves produce a protein known as GPR17, which blocks the first step of OPC differentiation into mature cells. Second, inflammatory cells in the injured nerves interfere with another step of OPC differentiation.
Combination treatment restores myelin
After testing a set of available compounds, co-first author Jing Wang, PhD, of the He lab, discovered that montelukast, an anti-inflammatory used for treatment of asthma and seasonal allergies, blocked development (or action) of GPR-17. Some axon remyelination was restored but only in about approximately 15 percent of treated nerve cells.
However, myelination rates were boosted significantly after removing immune cells, called microglia, from the damaged nerve cells with a drug called PLX3397. By itself, PLX3397 increased remyelination in 21 percent of axons. In combination with montelukast, the combination lead to remyelination in about 60 percent of damaged axons.
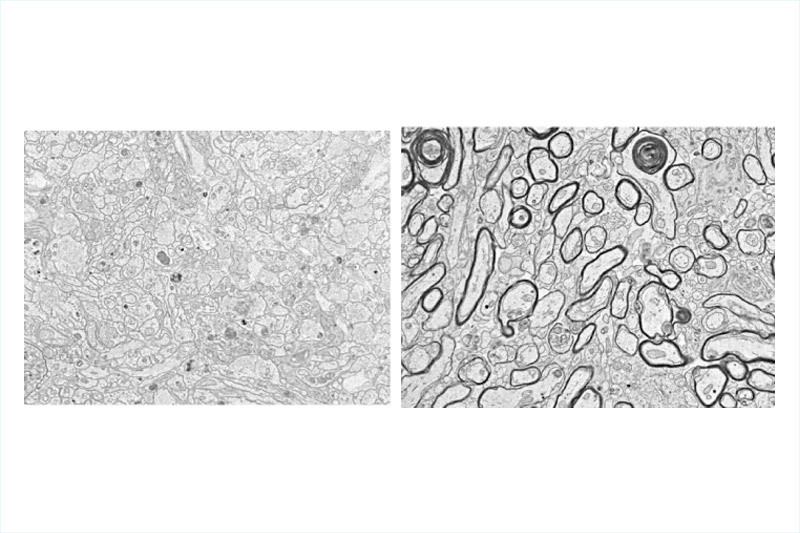
Typically, microglia act as scavengers in the central nervous system, looking for damaging inflammatory cells and infectious compounds and removing them from the site of the injury. However, in this case, the presence of microglia stopped the OPCs from developing into normal oligodendrocytes. Further studies with microglia showed that when they were removed later after injury — in this case two weeks after injury to the optic nerve — OPC development followed more normally.
“In a mice model of optic nerve injury, we found that when we combined treatment of montelukast and removing microglia later after injury, the majority of axons could be remyelinated,” He says.
Relevance to multiple sclerosis
In the advanced stage of multiple sclerosis (MS), injured nerves suffer from myelination failure. While this research was not conducted in a MS mouse model, the fact that He’s team discovered a way to remyelinate regenerated axons is encouraging since no currently available MS treatments currently work by rebuilding the myelin sheath.
“It is similar to what we see in the optic nerve after injury, “says He. “Both suffer from myelination failure so we believe our results could have some implications for new MS treatments, particularly for progressing MS.”
Other authors on this paper include co-first author Xuelian He, Huyan Meng, Yi Li, Phillip Dmitriev, Feng Tian, and Jessica C. Page of Boston Children’s, and Richard Lu of Cincinnati Children’s Hospital.
This study was supported by grant from the National Eye Institute, Dr. Miriam and Sheldon G. Adelson Medical Research Foundation, and the National Center for Complementary and Integrative Health.
Learn more about research from the F.M.Kirby Neurobiology Center.
Related Posts :
-

Scar-free healing after spinal cord injury relies on specialized cells
Scar tissue prevents nerves from communicating with each other. Microglia cells in the central nervous system help prevent scar tissue ...
-

Discoveries promise new strides for spinal cord injury patients
When neurobiologist Clifford Woolf, MB, BCh, PhD, began investigating potential treatments for spinal cord injury more than 30 years ago at ...
-

Promising advances in fetal therapy for vein of Galen malformation
In 2024, Megan Ingram* of California and her husband were preparing for the birth of their third child when a 34-week ...
-

The hidden burden of solitude: How social withdrawal influences the adolescent brain
Adolescence is a period of social reorientation: a shift from a world centered on parents and family to one shaped ...