How the new coronavirus gets into respiratory tissue — and may exploit one of our defenses
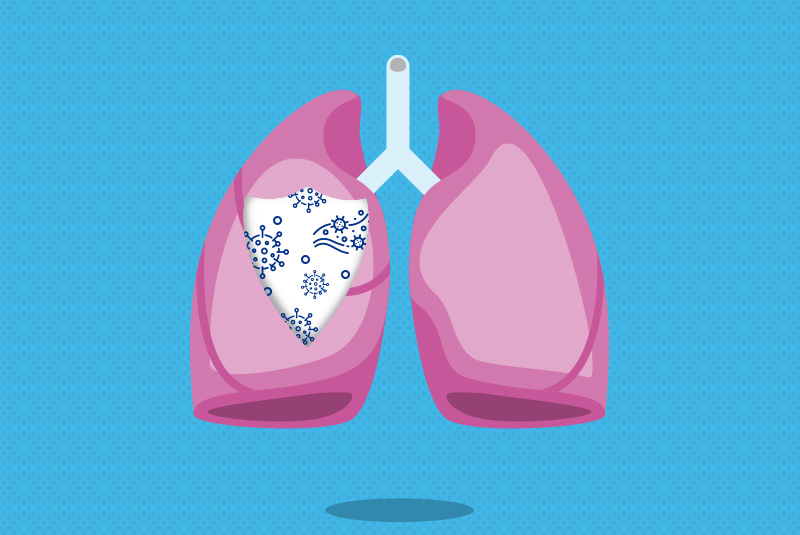
What makes SARS-CoV-2, the virus behind COVID-19, such a threat? A new study, led by Jose Ordovas-Montanes, PhD, at Boston Children’s Hospital and Alex K. Shalek, PhD, at MIT, pinpoints the likely cell types the virus infects. Unexpectedly, it also shows that one of the body’s main defenses against viral infections may actually help the virus infect those very cells. Findings were published this week by the journal Cell.
The peer-reviewed study, published as a preprint, will help focus efforts to understand what SARS-CoV-2 does in the body, why some people are more susceptible, and how best to search for treatments, the researchers say.
Multiple research models
When news broke about a new coronavirus in China, Ordovas-Montanes and Shalek had already been studying different cell types from throughout the human respiratory system and intestine. They also had gathered data from primates and mice.
In February, they began diving into these data.
“We started to look at cells from tissues such as the lining of the nasal cavity, the lungs, and gut, based on reported symptoms and where the virus has been detected,” says Ordovas-Montanes. “We wanted to provide the best information possible across our entire spectrum of research models.”

COVID-19-susceptible cells
Recent research had found that SARS-CoV-2 — like the closely related SARS-CoV that caused the SARS pandemic — uses a receptor called ACE2 to gain entry into human cells, aided by an enzyme called TMPRSS2. That led Ordovas-Montanes and Shalek and colleagues to ask a simple question. Which cells in respiratory and intestinal tissue express both ACE2 and TMPRSS2?
To get the answer, the team turned to single-cell RNA sequencing, which identifies which of roughly 20,000 genes are “on” in individual cells. They found that only a tiny percentage of human respiratory and intestinal cells, often well below 10 percent, make both ACE2 and TMPRSS2. Those cells fall into three types: goblet cells in the nose that secrete mucus; lung cells known as type II pneumocytes that help maintain the alveoli (the sacs where oxygen is taken in); and one type of so-called enterocytes that line the small intestine and are involved in nutrient absorption.
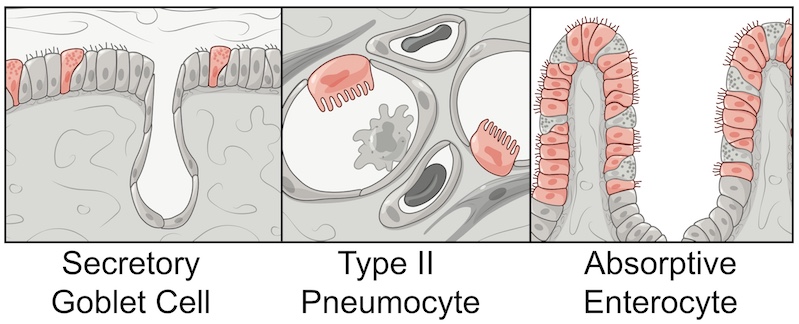
Sampling from non-human primates showed a similar pattern of susceptible cells.
“Many existing respiratory cell lines may not contain the full mix of cell types, and may miss the types that are relevant,” Ordovas-Montanes notes. “Once you understand which cells are infected, you can start to ask, ‘How do these cells work?’ ‘Is there anything within these cells that is critical for the virus’s life cycle?’ With more refined cellular models, we can perform better screens to find what existing drugs target that biology, providing a stepping stone to go into mice or non-human primates.”
Interferon: Helpful or harmful?
But it was the study’s second finding that most intrigues the scientists. They discovered that the ACE2 gene, which encodes the receptor SARS-CoV-2 uses to enter human cells, is stimulated by interferon — one of the body’s main defenses when it detects a virus. Interferon actually turned on the ACE2 gene at higher levels, potentially giving the virus new portals to get in.
“ACE2 is also critical in protecting people during various types of lung injury,” notes Ordovas-Montanes. “When ACE2 comes up, that’s usually a productive response. But since the virus uses ACE2 as a target, we speculate that it might be exploiting that normal protective response.”
Interferons, in fact, are being tested as a treatment for COVID-19. Would they help, or would they do more harm than good? That’s not yet clear.
“It might be that in some patients, because of the timing or the dose, interferon can contain the virus, while in others, interferon promotes more infection,” says Ordovas-Montanes. “We want to better understand where the balance lies, and how we can maintain a productive antiviral response without producing more target cells for the virus to infect.”
ACE inhibitors and cytokine storms
The findings may also raise new lines of inquiry around ACE inhibitors. These drugs are commonly used to treat hypertension, which has been linked to more severe COVID-19 disease. Are ACE inhibitors affecting people’s risk?
“ACE and ACE2 work in the same pathway, but they actually have different biochemical properties,” Ordovas-Montanes cautions. “It’s complex biology, but it will be important to understand the impact of ACE inhibitors on people’s physiological response to the virus.”
It’s also too soon to try to relate the study findings to the “cytokine storm,” a runaway inflammatory response that has been reported in very sick COVID-19 patients. Cytokines are a family of chemicals that rally the body’s immune responses to fight infections, and interferon is part of the family.
“It might be that we’re seeing a cytokine storm because of a failure of interferon to restrict the virus to begin with, so the lungs start calling for more help. That’s exactly what we’re trying to understand right now.”
Future directions
In addition, the team wants to explore what SARS-CoV-2 is doing in the cells it targets, and to study tissue samples from children and adults to understand why COVID-19 is typically less severe in younger people. Studies will continue at Boston Children’s with the support of Benjamin Raby, MD, MPH, chief of pulmonary medicine, Bruce Horwitz, MD, PhD, in emergency medicine, and Scott Snapper, MD, PhD, chief of gastroenterology.
Carly Ziegler, Samuel Allon, and Sarah Nyquist, of MIT and Harvard, and Ian Mbano of the Africa Health Research Institute were co-first authors on the paper in Cell. The study was done in collaboration with the Human Cell Atlas (HCA) Lung Biological Network group. The authors report no competing interests. See the paper for a full list of funders and authors.
“This has been an incredible community effort — not just within Boston, but also with collaborators around the world who have shared their unpublished data to try and make potentially relevant information available as rapidly as possible,” says Shalek, who was co-senior author on the paper with Ordovas-Montanes. “It’s inspiring to see how much can be accomplished when everyone comes together to tackle a problem.”
Explore Boston Children’s response to COVID-19 and more coronavirus research at the hospital.
Related Posts :
-

Four things you should know about MAPCAs treatment
As the first grandchild in her family, Hannah Homan is in demand for frequent visits. She was also the focus ...
-

Treating MAPCAs with unifocalization surgery and cardiology care
Children born with a rare form of tetralogy of Fallot (ToF) face a challenging type of congenital heart ...
-

A unique marker for pericytes could help forge a new path for pulmonary hypertension care
Pulmonary arterial hypertension (PAH) is a rare condition that’s difficult to treat. The hallmarks of the disease — narrowing of ...
-

A new druggable cancer target: RNA-binding proteins on the cell surface
In 2021, research led by Ryan Flynn, MD, PhD, and his mentor, Nobel laureate Carolyn Bertozzi, PhD, opened a new chapter ...