How families are reshaping Shwachman-Diamond syndrome research

No one knew the heartache about to unfold when Savannah and Brett Lillywhite first began thinking about having a family 10 years ago.
The Lillywhites
Savannah and Brett are both the unlikely carriers of a rare condition called Shwachman-Diamond syndrome — SDS for short — a blood disorder that can lead to bone marrow failure and increase the risk of cancer. The Lillywhites were unaware of the faulty gene that lurked in their DNA.
“I joke that we’re either soulmates, or we were never meant to be together,” says Savannah. “But I love him — so instead I say, we are soulmates meant to go through fire together.”
Their first child, Ethan, was born healthy. They had no way of knowing what was in store.

Then came Andrew.
“His life was fast and furious,” says Savannah. “He was diagnosed with SDS and passed away at 3 months. It was the loneliest time of our lives.”
Five years later, following a failed in vitro fertilization (IVF) attempt, the Lillywhites decided to test fate. The odds were on their side — or so they thought. “We couldn’t afford to go the genetic alteration route again,” says Savannah. “But we felt we were supposed to have another child and told ourselves, it will be fine. We played the genetic lottery and lost.”
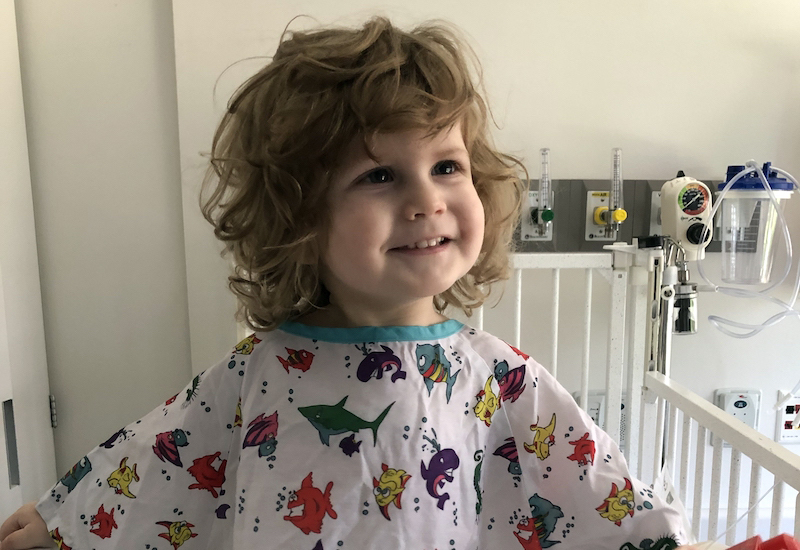
Lincoln Lillywhite was born with SDS on July 5, 2017, nearly five years to the day Andrew was born.
SDS Registry
This September, with 2-year-old Lincoln in tow, the Lillywhites flew from their home in Beaumont, Texas, to Boston. They were here to connect with other SDS families, many who embraced warmly as they spotted one another across the room. The connections run deep, even when a face is unfamiliar.
Eighty-five family members, all affected by SDS in one way or another, were here for SDS Family Day, an event hosted by the Shwachman-Diamond Syndrome Registry. Centered at Boston Children’s Hospital and Cincinnati Children’s Hospital Medical Center, the goal of the SDS registry is to fuel research through the collection of blood and bone marrow samples.
“Just learning about the registry is huge,” says Savannah. “— to know people care, to know there’s hope for Lincoln.”
Dr. Akiko Shimamura, an SDS researcher — who the Lillywhites describe as “the perfect trifecta: gentle, kind, brilliant” — is the director of the SDS registry. She’s also the director of the Bone Marrow Failure and Myelodysplastic Syndrome Program at Dana-Farber/Boston Children’s Cancer and Blood Disorders Center.
“Even if a child with SDS is doing well, they’re still at risk for complications,” Dr. Shimamura says. “So, part of the challenge is getting families and the medical community to recognize that these children need to be monitored regularly, because once the leukemia develops, treatments can be limited.”
The Gotshalls
Dr. Shimamura offers hope to families when there is none.
She did this for the Gotshalls in 2016 after Chase, then 9, was diagnosed with SDS, which developed into myelodysplastic syndrome, a rare disease of the blood, and rapidly progressed to acute myeloid leukemia (AML), a type of cancer with very low survival rates in children.
The relentless research began.
“It was all very shocking in a deer-in-the-headlights sort of way,” says Chase’s mom, Manda Gotshall. “It’s difficult to wrap your head around it. What do we do now? What is the next step?”
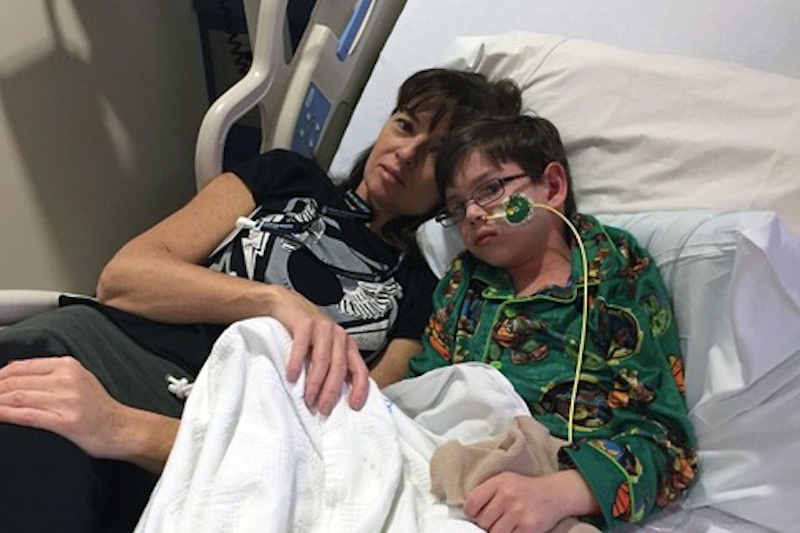
The next step was one of necessity, not choice.
Chase was placed on the waiting list for a bone marrow transplant. And although a suitable match was located shortly after, a new set of worries loomed.
“People with SDS have an increased sensitivity toward the chemotherapy used during the bone marrow transplant conditioning process,” says Dr. Shimamura. So, she began seeking alternatives for Chase.
A bone marrow failure clinical trial in Seattle showed promise — a chemotherapy medication with strong anti-tumor effects, but with lower toxicity than other agents available — however, Chase didn’t meet the criteria. Nevertheless, the data was promising in other high-risk cases.
“Chase could die if we did not get approval to try this,” says Dr. Shimamura. “The company provided the drug for free, and I petitioned the FDA, and they granted approval.”
Since the transplant, Chase has not had any signs of AML. “He is in remission,” says Dr. Shimamura. “He’s the rare AML case I know of with SDS who survived.”

Hope in the shadow of SDS
Manda is among the many family members, including the Lillywhites, who were at SDS Family Day. She says she is here to learn more about SDS, what her family can expect, and what’s in store.
“As Chase grows and his body starts to change, will he develop other things or is he in the clear?” she says. “— I feel like we’ll never be in the clear.”
Hope is on the horizon in the form of FDA approval for the chemotherapy drug used in Chase’s bone marrow transplant, and two new clinical trials are in development — one for bone marrow transplant diseases and the other for MDS and AML in SDS.
Lincoln Lillywhite was recently placed on the bone marrow transplant list, but Savannah is hopeful, even in the face of overwhelming odds.
“I have this amazing support system,” she says. “The first time it was a sprint, and this time, it’s going to be a long journey. Knowing I have the SDS community behind me makes me feel much more empowered and strengthened going into round two.”
Learn more about the SDS registry.
Related Posts :
-

The dopamine reset: Restoring what’s missing in AADC deficiency
In March 2023, a young girl came to Boston Children’s Hospital unable to hold up her head — one striking symptom ...
-

Parsing the promise of inosine for neurogenic bladder
Spinal cord damage — whether from traumatic injury or conditions such as spina bifida — can have a profound impact on bladder ...
-

Thanks to Carter and his family, people are talking about spastic paraplegia
Nine-year-old Carter may be the most devoted — and popular — sports fan in his Connecticut town. “He loves all sports,” ...
-

Unveiling the hidden impact of moyamoya disease: Brain injury without symptoms
Moyamoya disease — a rare, progressive condition that narrows the brain’s blood vessels — leads to an increased risk of stroke ...